Publications
A surface lipoprotein on Pasteurella multocida binds complement factor I to promote immune evasion
PLOS Pathogens
Nguyen QH, Lai CHR, Norris MJ, Ng D, Shah M, et al. (2025) A surface lipoprotein on Pasteurella multocida binds complement factor I to promote immune evasion. PLOS Pathogens 21(5): e1012686. https://doi.org/10.1371/journal.ppat.1012686
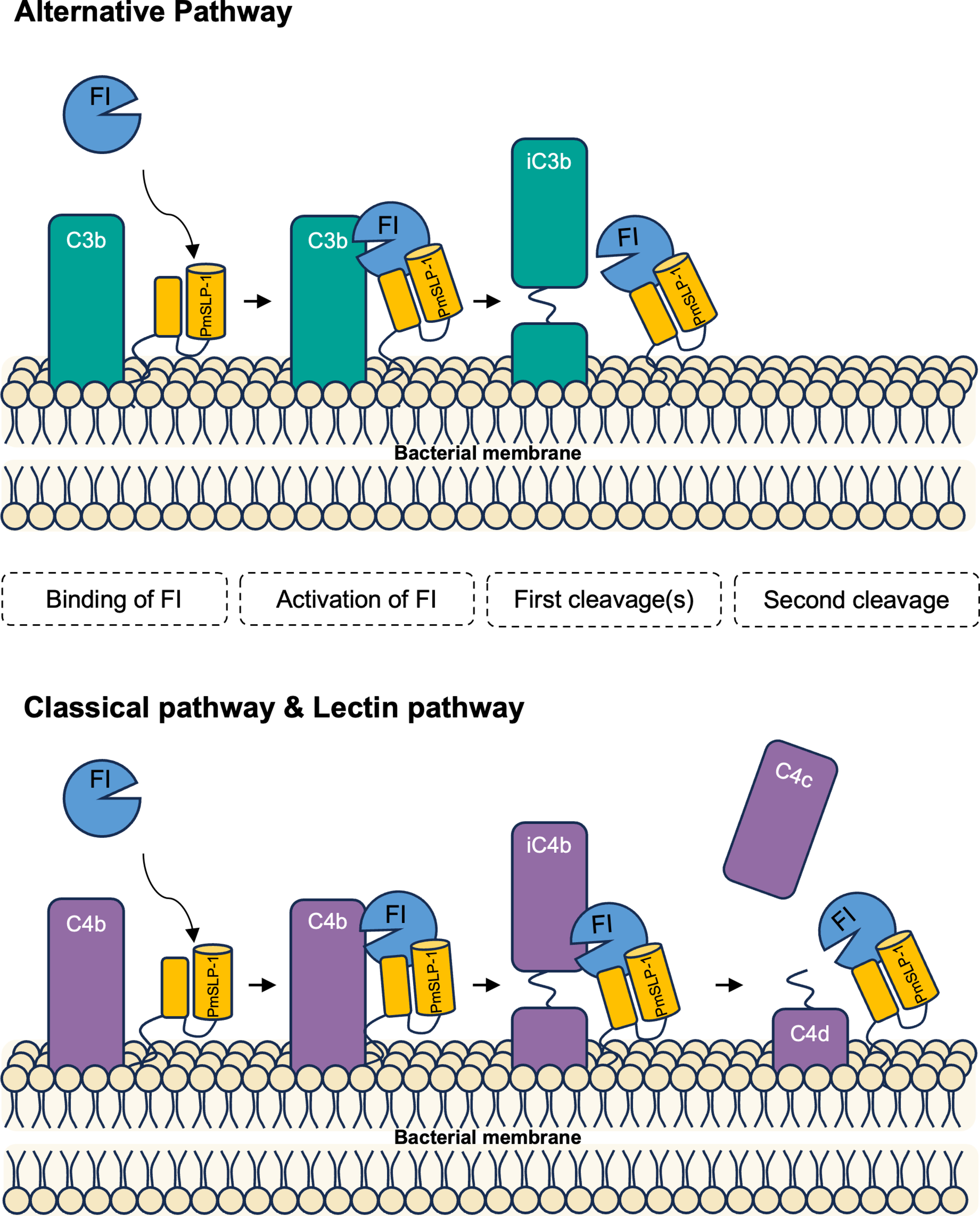
Pasteurella multocida is the leading cause of wound infections in humans following animals’ bites or scratches. This bacterium is also commonly found in the respiratory tract of many mammals and can cause serious diseases resulting in the rapid death of infected animals, especially cattle. To prevent these infections in cattle, a subunit-based vaccine utilizing the surface lipoprotein PmSLP was developed and showed remarkable protection with a single dose administration. Here, we report that PmSLP binds host complement factor I (FI) and facilitates cleavage of complement components C3b and C4b independently of any cofactors (e.g., FH, C4BP), thereby allowing the pathogen to evade host defence. Cryo-EM structure of PmSLP bound to FI reveals that PmSLP stimulates FI enzymatic activity by stabilizing the catalytic domain. This is the first time that a bacterial protein has been shown to directly activate FI independent of complement cofactors and target all arms of the complement cascade.
Slam is an outer membrane protein that is required for the surface display of lipidated virulence factors in Neisseria
Nature Microbiology
Hooda, Y., Lai, CL., Judd, A. et al. Slam is an outer membrane protein that is required for the surface display of lipidated virulence factors in Neisseria. Nat Microbiol 1, 16009 (2016). https://doi.org/10.1038/nmicrobiol.2016.9
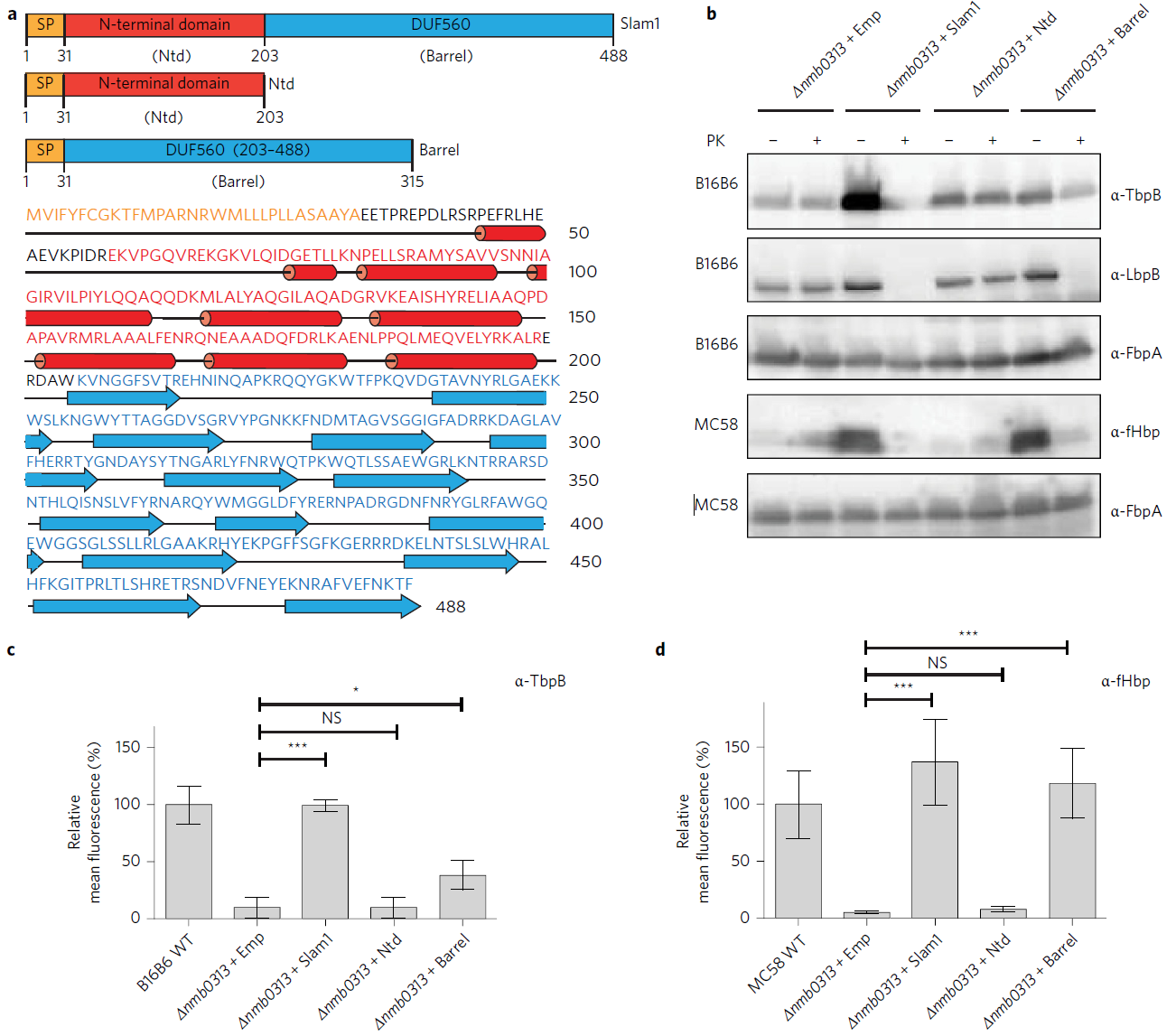
Lipoproteins decorate the surface of many Gram-negative bacterial pathogens, playing essential roles in immune evasion and nutrient acquisition. In Neisseria spp., the causative agents of gonorrhoea and meningococcal meningitis, surface lipoproteins (SLPs) are required for virulence and have been extensively studied as prime candidates for vaccine development. However, the machinery and mechanism that allow for the surface display of SLPs are not known. Here, we describe a transposon (Tn5)-based search for the proteins required to deliver SLPs to the surface of Neisseria meningitidis, revealing a family of proteins that we have named the surface lipoprotein assembly modulator (Slam). N. meningitidis contains two Slam proteins, each exhibiting distinct substrate preferences. The Slam proteins are sufficient to reconstitute SLP transport in laboratory strains of Escherichia coli, which are otherwise unable to efficiently display these lipoproteins on their cell surface. Immunoprecipitation and domain probing experiments suggest that the SLP, TbpB, interacts with Slam during the transit process; furthermore, the membrane domain of Slam is sufficient for selectivity and proper surface display of SLPs. Rather than being a Neisseria-specific factor, our bioinformatic analysis shows that Slam can be found throughout proteobacterial genomes, indicating a conserved but until now unrecognized virulence mechanism.
The structural basis of transferrin sequestration by transferrin-binding protein B
Nature Structural & Molecular Biology
Calmettes, C., Alcantara, J., Yu, RH. et al. The structural basis of transferrin sequestration by transferrin-binding protein B. Nat Struct Mol Biol 19, 358–360 (2012). https://doi.org/10.1038/nsmb.2251
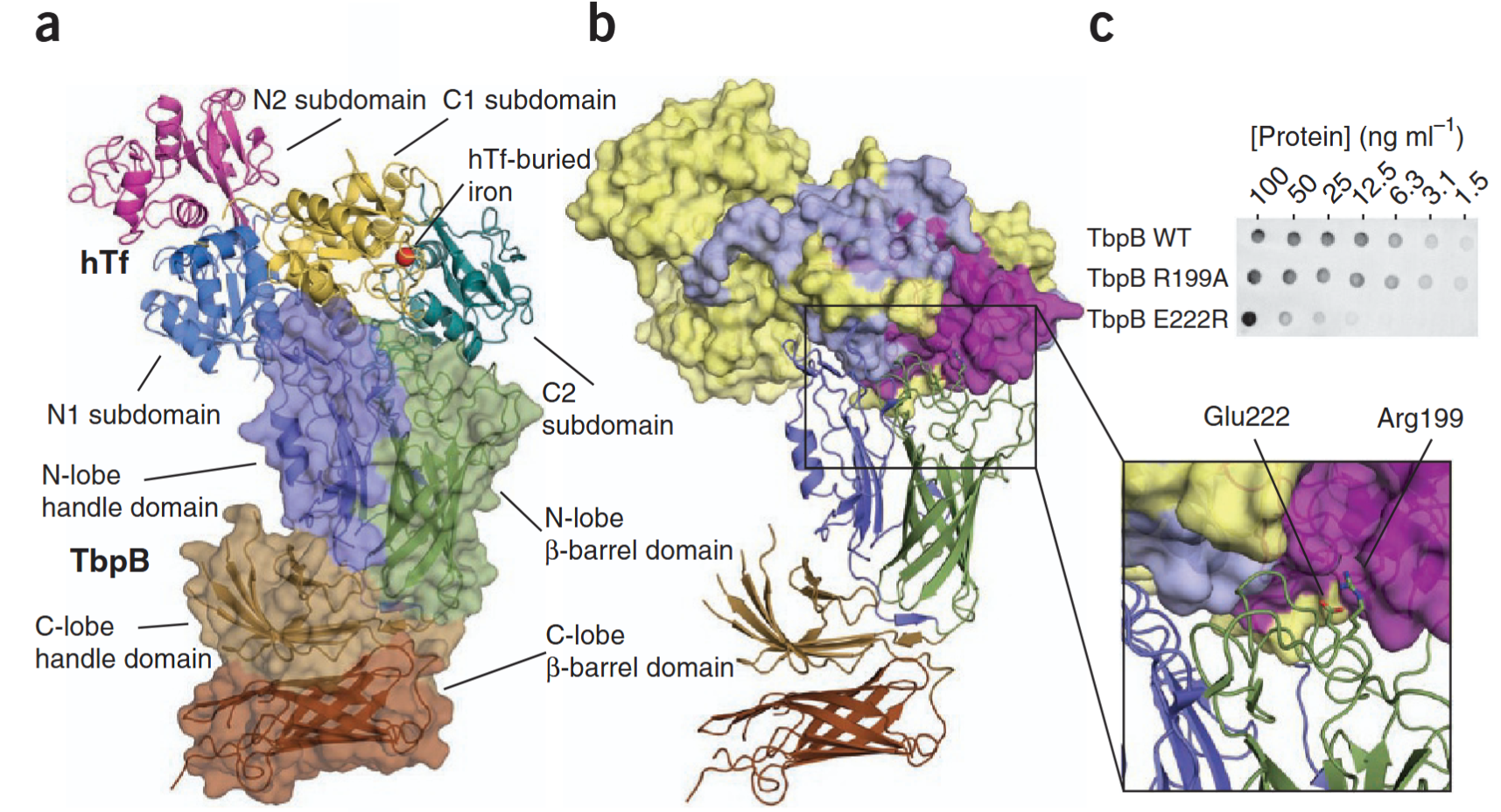
Neisseria meningitidis, the causative agent of bacterial meningitis, acquires the essential element iron from the host glycoprotein transferrin during infection through a surface transferrin receptor system composed of proteins TbpA and TbpB. Here we present the crystal structures of TbpB from N. meningitidis in its apo form and in complex with human transferrin. The structure reveals how TbpB sequesters and initiates iron release from human transferrin.
Rational selection of TbpB variants yields a bivalent vaccine with broad coverage against Neisseria gonorrhoeae
NPJ Vaccines
Fegan, J.E., Islam, E.A., Curran, D.M. et al. Rational selection of TbpB variants yields a bivalent vaccine with broad coverage against Neisseria gonorrhoeae. npj Vaccines 10, 10 (2025). https://doi.org/10.1038/s41541-024-01054-0
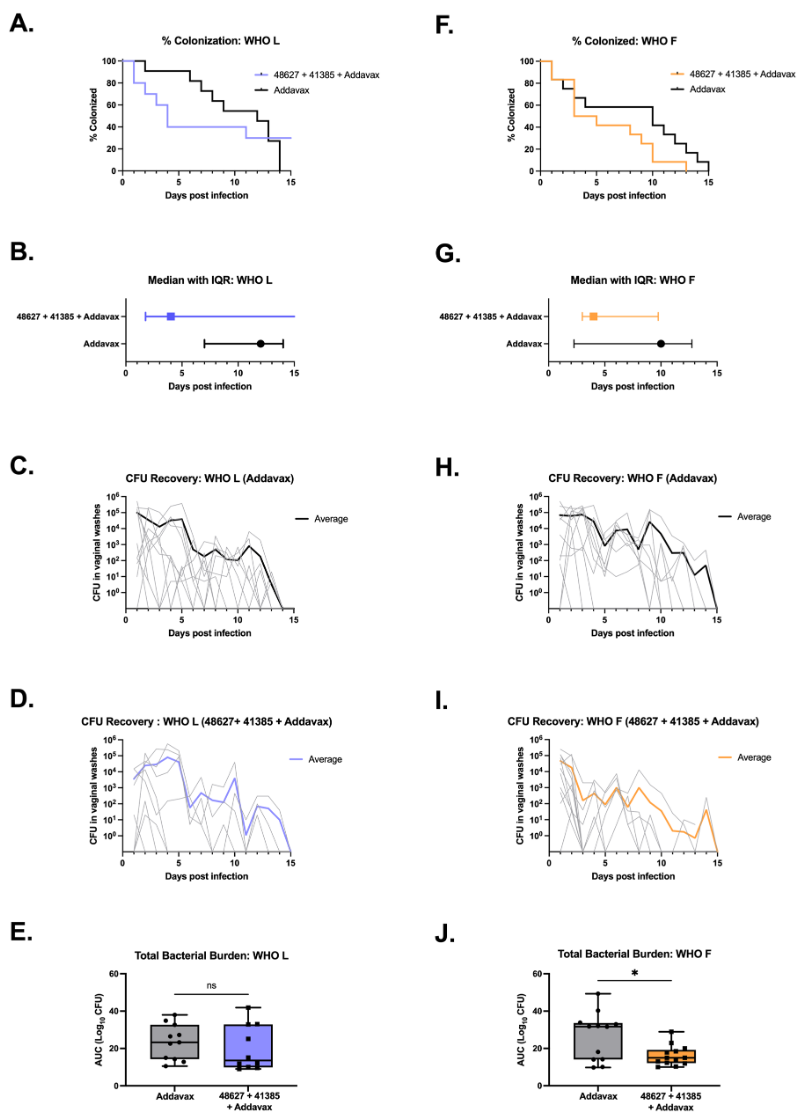
Neisseria gonorrhoeae is an on-going public health problem due in part to the lack of success with efforts to develop an efficacious vaccine to prevent this sexually transmitted infection. The gonococcal transferrin binding protein B (TbpB) is an attractive candidate vaccine antigen. However, it exhibits high levels of antigenic variability, posing a significant obstacle in evoking a broadly protective immune response. Here, we utilize phylogenetic information to rationally select TbpB variants for inclusion into a gonococcal vaccine and identify two TbpB variants that together elicit a highly cross-reactive antibody response against a diverse panel of TbpB variants and clinically relevant gonococcal strains. This formulation performed well in experimental proxies of real-world usage, including eliciting bactericidal activity against diverse gonococcal strains and decreasing the median duration of colonization after vaginal infection in female mice. These data support the use of a combination of TbpB variants for a broadly protective gonococcal vaccine.
Reconstitution of surface lipoprotein translocation through the Slam translocon
eLife Sciences
Minh Sang Huynh, Yogesh Hooda, Yuzi Raina Li, Maciej Jagielnicki, Christine Chieh-Lin Lai, Trevor F Moraes (2022) Reconstitution of surface lipoprotein translocation through the Slam translocon eLife 11:e72822 https://doi.org/10.7554/eLife.72822
Surface lipoproteins (SLPs) are peripherally attached to the outer leaflet of the outer membrane in many Gram-negative bacteria, playing significant roles in nutrient acquisition and immune evasion in the host. While the factors that are involved in the synthesis and delivery of SLPs in the inner membrane are well characterized, the molecular machinery required for the movement of SLPs to the surface are still not fully elucidated. In this study, we investigated the translocation of a SLP TbpB through a Slam1-dependent pathway. Using purified components, we developed an in vitro translocation assay where unfolded TbpB is transported through Slam1-containing proteoliposomes, confirming Slam1 as an outer membrane translocon. While looking to identify factors to increase translocation efficiency, we discovered the periplasmic chaperone Skp interacted with TbpB in the periplasm of Escherichia coli. The presence of Skp was found to increase the translocation efficiency of TbpB in the reconstituted translocation assays. A knockout of Skp in Neisseria meningitidis revealed that Skp is essential for functional translocation of TbpB to the bacterial surface. Taken together, we propose a pathway for surface destined lipoproteins, where Skp acts as a holdase for Slam-mediated TbpB translocation across the outer membrane.
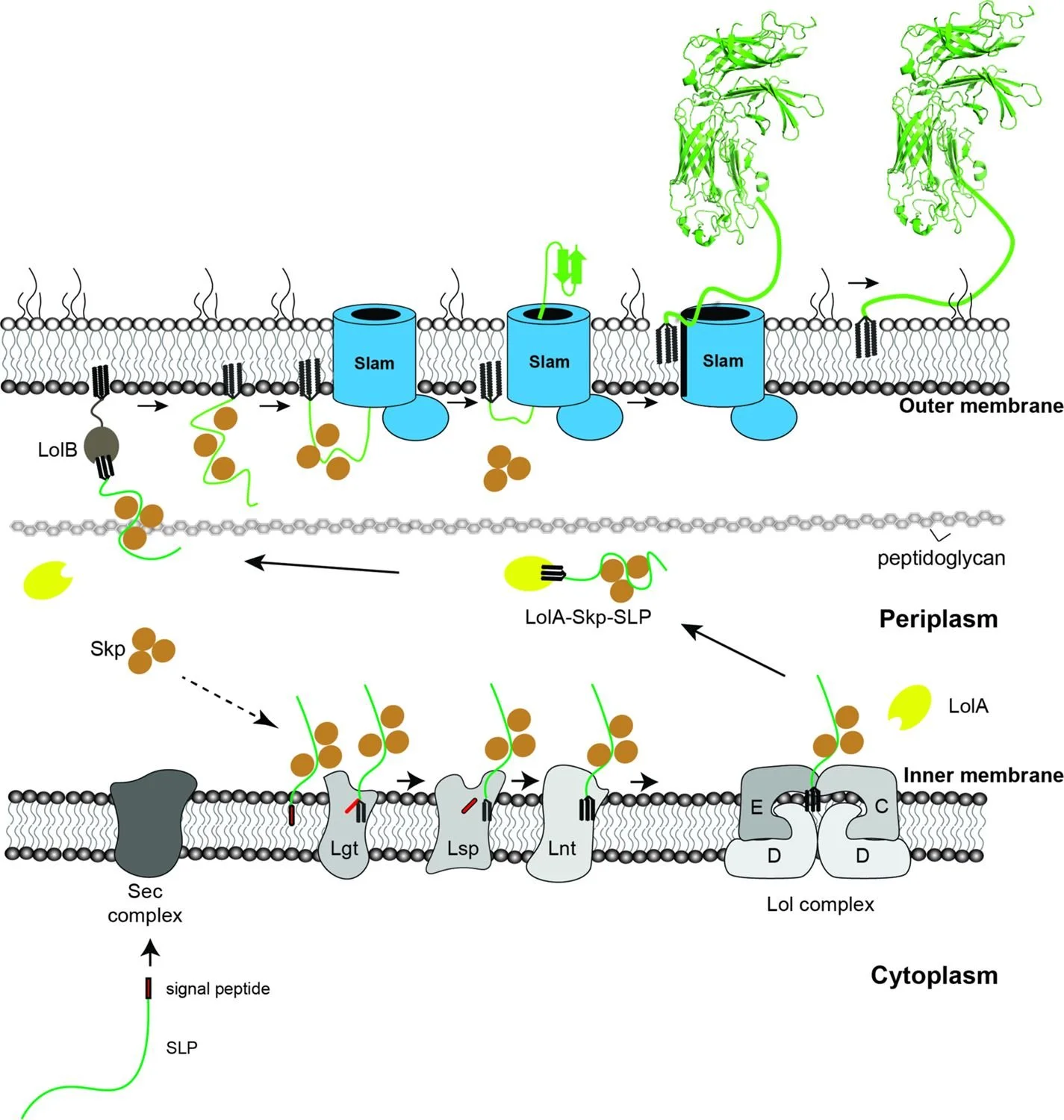
Reverse vaccinology-based identification of a novel surface lipoprotein that is an effective vaccine antigen against bovine infections caused by Pasteurella multocida
PLOS Pathogens
Islam EA, Fegan JE, Tefera TA, Curran DM, Waeckerlin RC, et al. (2023) Reverse vaccinology-based identification of a novel surface lipoprotein that is an effective vaccine antigen against bovine infections caused by Pasteurella multocida. PLOS Pathogens 19(3): e1011249. https://doi.org/10.1371/journal.ppat.1011249
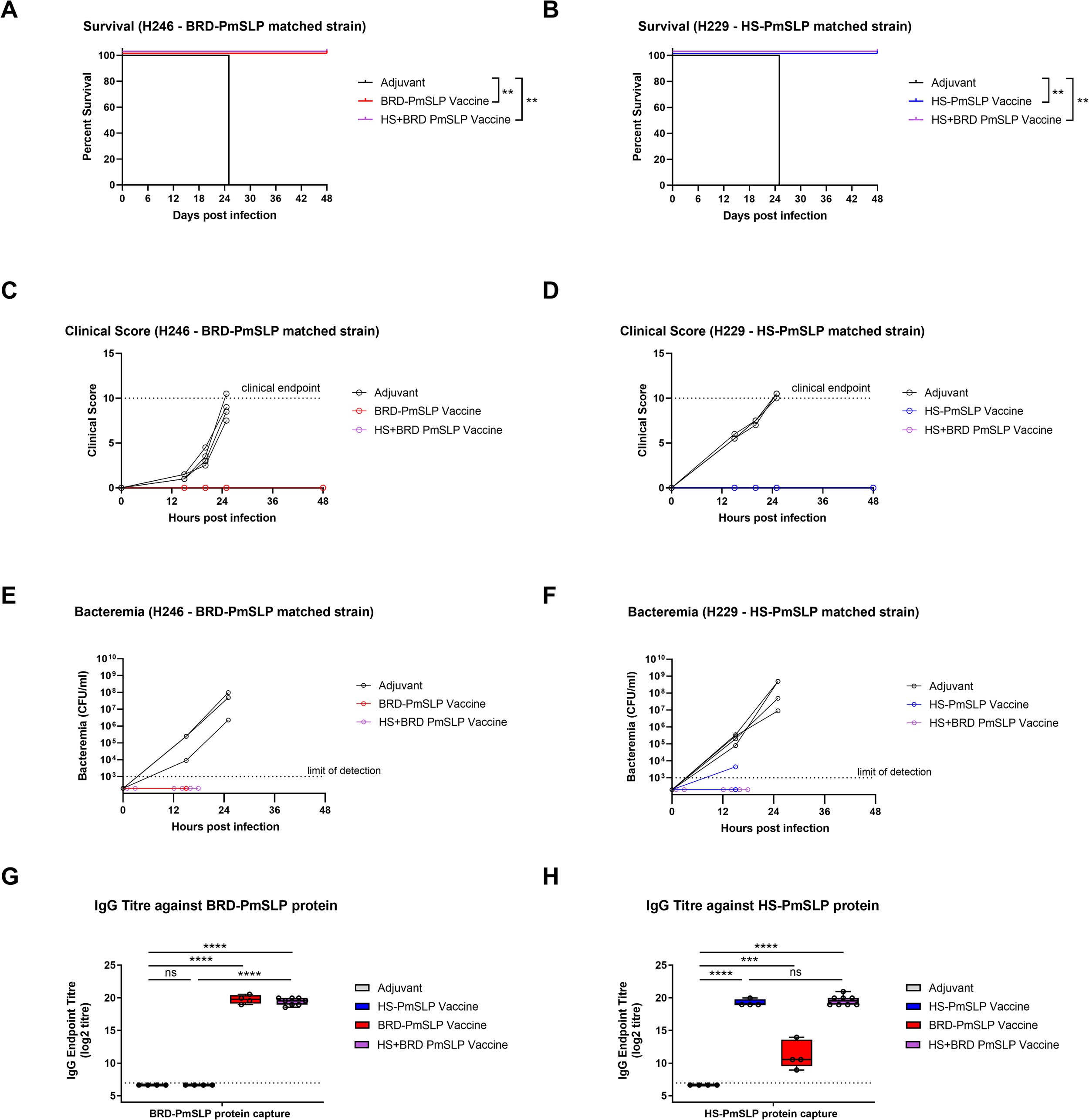
Pasteurella multocida can infect a multitude of wild and domesticated animals, with infections in cattle resulting in hemorrhagic septicemia (HS) or contributing to bovine respiratory disease (BRD) complex. Current cattle vaccines against P. multocida consist of inactivated bacteria, which only offer limited and serogroup specific protection. Here, we describe a newly identified surface lipoprotein, PmSLP, that is present in nearly all annotated P. multocida strains isolated from cattle. Bovine associated variants span three of the four identified phylogenetic clusters, with PmSLP-1 and PmSLP-2 being restricted to BRD associated isolates and PmSLP-3 being restricted to isolates associated with HS. Recombinantly expressed, soluble PmSLP-1 (BRD-PmSLP) and PmSLP-3 (HS-PmSLP) vaccines were both able to provide full protection in a mouse sepsis model against the matched P. multocida strain, however no cross-protection and minimal serum IgG cross-reactivity was identified. Full protection against both challenge strains was achieved with a bivalent vaccine containing both BRD-PmSLP and HS-PmSLP, with serum IgG from immunized mice being highly reactive to both variants. Year-long stability studies with lyophilized antigen stored under various temperatures show no appreciable difference in biophysical properties or loss of efficacy in the mouse challenge model. PmSLP-1 and PmSLP-3 vaccines were each evaluated for immunogenicity in two independent cattle trials involving animals of different age ranges and breeds. In all four trials, vaccination with PmSLP resulted in an increase in antigen specific serum IgG over baseline. In a blinded cattle challenge study with a recently isolated HS strain, the matched HS-PmSLP vaccine showed strong efficacy (75–87.5% survival compared to 0% in the control group). Together, these data suggest that cattle vaccines composed of PmSLP antigens can be a practical and effective solution for preventing HS and BRD related P. multocida infections.
Recent Publications
A pore-forming antiphage defence is activated by oligomeric phage proteins
Patel PH, McCarthy MR, Taylor VL, Cole GB, Zhang C, Edghill MM, Getz LJ, Fung BCM, Moraes TF, Davidson AR, Norris MJ, Maxwell KL. A pore-forming antiphage defence is activated by oligomeric phage proteins. Nature. 2026 Feb 4. doi: 10.1038/s41586-025-10075-1. Epub ahead of print. PMID: 41639445.
Structure-based engineering of a nutrient acquisition protein enhances neutralizing antibodies and protection for the development of a gonococcal vaccine
Au NYT, Zhong EZL, Fegan JE, Islam EA, Xu SX, Ng D, Shah M, Caruso LL, Lai CCL, Schryvers AB, Gray-Owen SD, Moraes TF. Structure-based engineering of a nutrient acquisition protein enhances neutralizing antibodies and protection for the development of a gonococcal vaccine. bioRxiv [Preprint]. 2025 Nov 8:2025.11.06.685839. doi: 10.1101/2025.11.06.685839. PMID: 41279537; PMCID: PMC12637532.
A surface lipoprotein on Pasteurella multocida binds complement factor I to promote immune evasion
Nguyen QH, Lai CHR, Norris MJ, Ng D, Shah M, Lai CC, Isenman DE, Moraes TF. A surface lipoprotein on Pasteurella multocida binds complement factor I to promote immune evasion. PLoS Pathog. 2025 May 6;21(5):e1012686. doi: 10.1371/journal.ppat.1012686. PMID: 40327719; PMCID: PMC12080921.
Adjuvant-dependent impacts on vaccine-induced humoral responses and protection in preclinical models of nasal and genital colonization by pathogenic Neisseria
Islam EA, Fegan JE, Zeppa JJ, Ahn SK, Ng D, Currie EG, Lam J, Moraes TF, Gray-Owen SD. Adjuvant-dependent impacts on vaccine-induced humoral responses and protection in preclinical models of nasal and genital colonization by pathogenic Neisseria. Vaccine. 2025 Feb 27;48:126709. doi: 10.1016/j.vaccine.2025.126709. Epub 2025 Jan 15. PMID: 39817984.
Rational selection of TbpB variants yields a bivalent vaccine with broad coverage against Neisseria gonorrhoeae
Fegan JE, Islam EA, Curran DM, Ng D, Au NYT, Currie EG, Zeppa JJ, Lam J, Schryvers AB, Moraes TF, Gray-Owen SD. Rational selection of TbpB variants yields a bivalent vaccine with broad coverage against Neisseria gonorrhoeae. NPJ Vaccines. 2025 Jan 15;10(1):10. doi: 10.1038/s41541-024-01054-0. PMID: 39814726; PMCID: PMC11736018.
Investigating the importance of selected surface-exposed loops in HpuB for hemoglobin binding and utilization by Neisseria gonorrhoeae
Awate OA, Ng D, Stoudenmire JL, Moraes TF, Cornelissen CN. Investigating the importance of selected surface-exposed loops in HpuB for hemoglobin binding and utilization by Neisseria gonorrhoeae. Infect Immun. 2024 Jul 11;92(7):e0021124. doi: 10.1128/iai.00211-24. Epub 2024 Jun 12. PMID: 38864605; PMCID: PMC11238557.
POTRA domains of the TamA insertase interact with the outer membrane and modulate membrane properties
Mellouk A, Jaouen P, Ruel LJ, Lê M, Martini C, Moraes TF, El Bakkouri M, Lagüe P, Boisselier E, Calmettes C. POTRA domains of the TamA insertase interact with the outer membrane and modulate membrane properties. Proc Natl Acad Sci U S A. 2024 Jul 9;121(28):e2402543121. doi: 10.1073/pnas.2402543121. Epub 2024 Jul 3. PMID: 38959031; PMCID: PMC11252910.
Prevalence of Slam-dependent hemophilins in Gram-negative bacteria
Shin HE, Pan C, Curran DM, Bateman TJ, Chong DHY, Ng D, Shah M, Moraes TF. Prevalence of Slam-dependent hemophilins in Gram-negative bacteria. J Bacteriol. 2024 Jun 20;206(6):e0002724. doi: 10.1128/jb.00027-24. Epub 2024 May 30. PMID: 38814789; PMCID: PMC11332172.
The DIAPH3 linker specifies a β-actin network that maintains RhoA and Myosin-II at the cytokinetic furrow
Shah R, Panagiotou TC, Cole GB, Moraes TF, Lavoie BD, McCulloch CA, Wilde A. The DIAPH3 linker specifies a β-actin network that maintains RhoA and Myosin-II at the cytokinetic furrow. Nat Commun. 2024 Jun 19;15(1):5250. doi: 10.1038/s41467-024-49427-2. PMID: 38897998; PMCID: PMC11187180.